中文
Yaogu New Talent | Lishan Biotech: Developing Innovative Drugs with Global Innovative Swarming Technology
In order to highlight the innovation and vitality of Zhangjiang's life science industry, the WeChat official account of "Zhangjiang Pharmaceutical Valley" launched the column of "New Talents of Pharmaceutical Valley", and invited new enterprises of Zhangjiang Pharmaceutical Valley to have in-depth dialogue and decode innovation. In this issue, we will have a conversation with Dr. William Chen, Chairman of Shanghai Lishan Biopharmaceutical Co., Ltd. (hereinafter referred to as "Lishan Biotech"), to learn how the company has embarked on a path of cross-border integration and innovation.

William Chen, Chairman of Lishan Biotechnology
At the National Finals of the 3rd National Postdoctoral Innovation and Entrepreneurship Competition, which concluded in October 2025, Dr.William Chen, Chairman of Lishan Biotech, led the "Intestinal Microbial Live Biopharmaceutical Treatment for Crohn's Disease" project, which stood out among many top teams in the biopharmaceutical field and won the "Silver Award".
When it comes to the key to this award, William Chen attributed it to the unique positioning and huge market potential of the project. He said, "We are committed to developing 'live biopharmaceuticals' derived from the human body itself, which provides a new, safe and effective solution for the treatment of difficult diseases such as Crohn's disease." Lishan Biotech focuses on AI empowered innovative drug research and development, with cutting-edge microbiological research as the fulcrum, committed to regulating the ecological balance of the human body through precision medicine and solving the unmet clinical needs of complex diseases.
Cross border integration: taking an unconventional path, specifically targeting the 'hard bones' to tackle
From the study of bacterial dynamics at Brown University, to the interdisciplinary exploration of microbiology and immunology at Einstein College of Medicine, and to the founding of Lishan Biotech upon returning to China, William Chen has always been at the forefront of "non consensus" scientific research. When most researchers focus on the "omics" aspect of microbiology, they choose to re understand the interaction mechanism between microorganisms and hosts from the physical perspective of bacterial movement and fluid mechanics, and thus construct a new paradigm for drug development.
During his doctoral research, under the joint training of Professor Jay X. Tang, a bacterial dynamics expert at Brown University, and Professor Sridhar Mani, an immunology and microbiology expert at Einstein College of Medicine, William Chen and his team made a breakthrough in the development of drugs for treating intestinal diseases from the perspectives of bacterial dynamics and fluid dynamics.
This systematic academic training spanning biophysics and biomedical fields has laid a solid foundation for its future interdisciplinary integration of AI and microbial drug research.

(Image source: Lishan Biotech)
In early 2023, Chen Weijie founded Lishan Biotechnology, embarking on an entrepreneurial journey from "deepening scientific research" to "industrialization of technology", continuously developing core technologies of "AI+live biopharmaceuticals", anchoring complex diseases such as Crohn's Disease (CD), Renal Cell Carcinoma (RCC), Ulcerative Colitis (UC), Autism Spectrum Disorder (ASD), Acute Leukemia (AL), and Alzheimer's disease (AD), and promoting the development and implementation of six cutting-edge pipelines.
Continuous innovation is a wider and deeper 'moat'
As an emerging therapy, live biopharmaceuticals are a "blue ocean" in the field of innovative drugs. There is no path to industrialization from the laboratory to the market that can be learned from. Lishan Biotech wants to find its own innovative development path in this field, which requires solving two major challenges: the difficulty of gut microbiota colonization and finding suppliers that meet the requirements of live bacterial fermentation and production.
01: Solving the difficulty of intestinal microbiota colonization
For solving the core technical difficulty of "difficult colonization of gut microbiota": the difficulty of foreign beneficial bacteria to survive, reproduce, and occupy a certain "ecological niche" in the human gut for a long time, ultimately unable to sustainably regulate the gut microbiota or have therapeutic effects. In short, it means that 'good bacteria that are ingested cannot be retained or rooted'.
Lishan Biotech has overcome this challenge with the world's first Swarming (swarm movement) technology. Swarming is a bacterial motility phenotype characterized by collective cooperative migration behavior, accompanied by adaptive changes in bacterial morphology and metabolic pathways, enabling efficient migration, colonization, and functional expression. This technology has demonstrated unique advantages in the fields of gut microbiota therapy and biofilm regulation, becoming a key means for Lishan Biology to overcome traditional colonization challenges. At present, this technology has obtained national invention patent authorization and simultaneously applied for PCT patents. According to the search results of the Chinese Academy of Sciences, it has reached the leading domestic and international advanced levels.
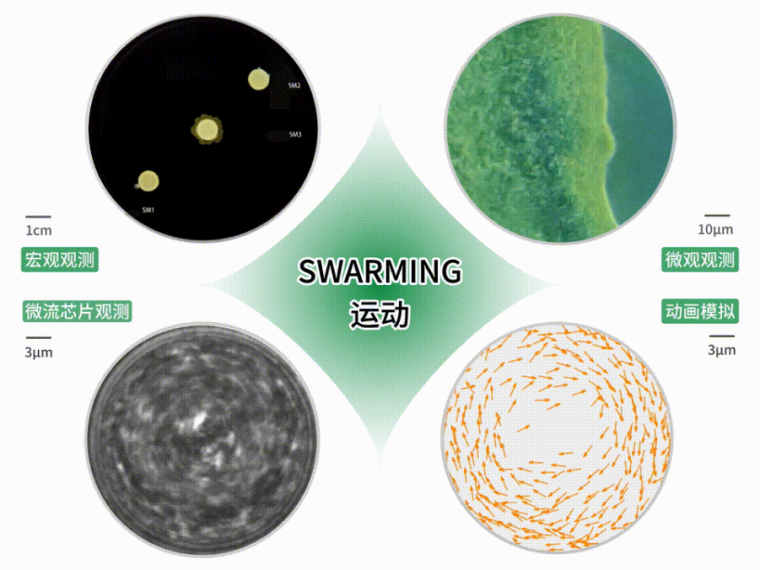
Multiple observation results of Swarming motion
The live biopharmaceutical developed by Lishan Biotech for Crohn's disease uses strains with Swarming phenotype. Animal experiments have confirmed that this strain has a significantly higher colonization rate at the site of intestinal ulcers through Swarming movement in a mouse model of Crohn's disease than ordinary probiotics. It can also form a biofilm protective layer on the surface of intestinal ulcers, covering and repairing damaged intestinal mucosa, demonstrating superior therapeutic potential.
02: Find suppliers that meet the requirements for live bacterial fermentation and production
Another challenge is to find suppliers that meet the requirements of live bacterial fermentation and production: due to the lack of industry standards and effective standard guidance, Chen Weijie led the team to boldly break through and did not rashly choose to cooperate with mature pharmaceutical companies. Instead, they formed a strategic alliance with a professional start-up supplier in a vertical field, aiming to jointly overcome process difficulties and develop industry standards.
At present, Lishan Biotech has completed laboratory establishment, strain isolation and identification, and biochemical and physical property testing of strains. The strain library has achieved fruitful results, successfully isolating over thousands of strains that can be industrialized, and building a production process system that meets GMP standards through external cooperation.
As the 'ammunition depot' for source innovation, these strains provide diversified target objects for production process development. The metabolic characteristics, stability, and efficacy mechanisms of each strain drive our in-depth research and development in optimizing culture medium formulations, precise regulation of fermentation parameters, and specific design of harvesting processes, thereby achieving the goal of improving product quality and forming a closed-loop mapping relationship of 'strain characteristics process parameters product quality'
At present, Lishan Biotechnology's Crohn's disease pipeline has completed Phase I clinical trials, and multiple follow-up pipelines are also progressing in an orderly manner according to plan, demonstrating the company's strong research and development momentum and future potential.
Integrating industry, academia, and research to empower rapid industrial development
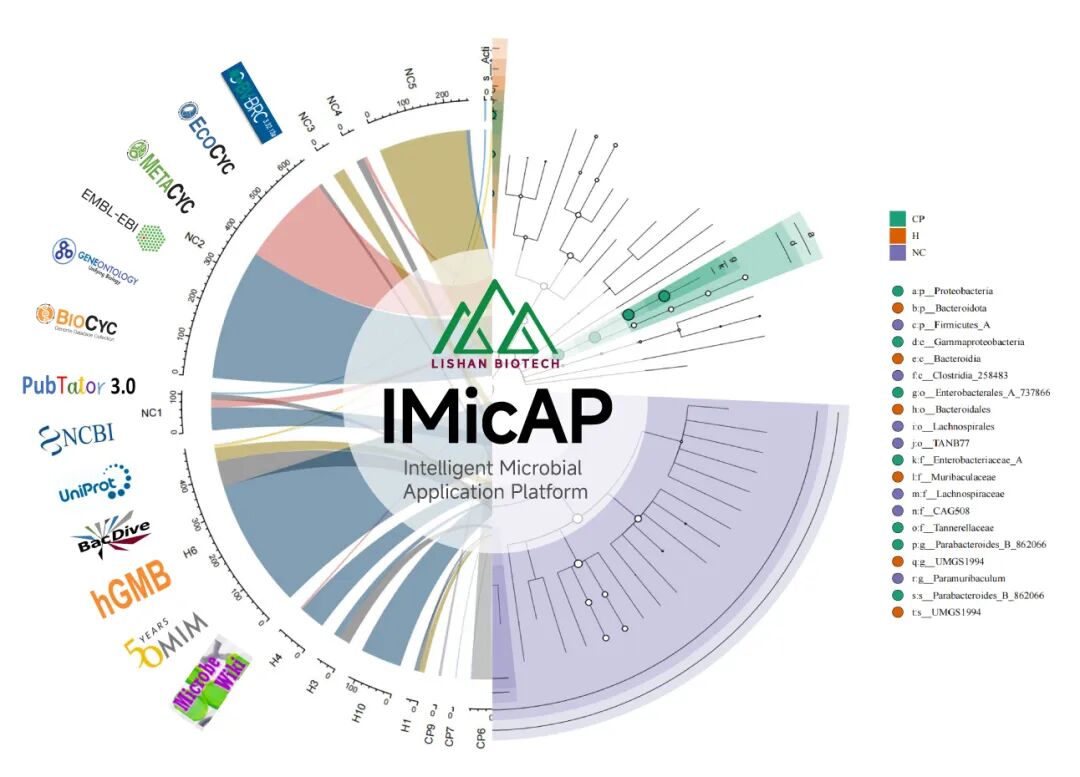
(Image source: Lishan Biotech)
While focusing on innovative research and development, Lishan Biotech has also explored a path of integrated development of industry, academia, research, and medicine. In August 2023, Lishan Biotech and the Institute of Intelligent Medicine at Fudan University jointly established the Intelligent Microbiome School Enterprise Joint Laboratory and independently developed the Intelligent Microbial Application Platform (IMicAP).
The IMicAP platform relies on the knowledge support of the Precision Medicine Knowledge Base of the Intelligent Medical College and the data support of affiliated hospitals. It integrates functions such as "cross platform knowledge query and natural language Q&A", "integrated data display and simple bioinformatics analysis", "forward-looking knowledge reasoning and rapid drug screening", and "precise disease risk prediction and intestinal microbiota matching". It will improve the efficiency of knowledge query, learning, and organization for researchers, pharmaceutical R&D personnel, and clinical doctors in the field of microbiology, provide "zero code" bioinformatics analysis, scientific research graphics, and forward-looking live biopharmaceutical screening and prediction, achieve accurate disease risk prediction based on intestinal microbiota detection, donor matching, and intelligent screening of innovative drugs for clinical inclusion.
People who make medicine ultimately have emotions. Only by maintaining their true colors and practicing their internal skills, will you eventually have the opportunity that belongs to you. With this belief, William Chen is leading his team to make breakthroughs in technology, process, and production, and accelerating the commercialization process of products with down-to-earth efforts.
Located in Zhangjiang Pharmaceutical Valley, the full process collaboration from laboratory to clinical can be achieved within a radius of 10 kilometers, with significant efficiency advantages and cost-effectiveness. Relying on a comprehensive biopharmaceutical industry chain and a talent pool and concentration in the biopharmaceutical field, Lishan Biotech will continue to integrate innovation and explore the "star sea" of life sciences.


